Making a Mouse a Human: Human‑Like Tuberculosis Pathology in Mice Without the ROS/RNS Killing Myth
Professor. William R Jacobs Jr*
*Department of Microbiology and Immunology, Albert Einstein College of Medicine, Morris Park Avenue, Bronx, NY, USA
*Corresponding author: Professor. William R Jacobs Jr., Dept. of Microbiology and Immunology, Albert Einstein College of Medicine, 1301 Morris Park Avenue, Bronx, NY 10461, USA, Telephone: +1 718-678-1075; Email: william.jacobs@einsteinmed.edu
Received: 12 December 2025; Revised: 04 February 2026; Accepted: 12 February 2026; Published: 17 February 2026
Copyright: © 2026 Jacobs Jr WR. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Professor. Jacobs Jr WR (2026) The Price of Protection Revisited: Weinrick’s Nos2?/? Model and Kramnik’s Genetic Blueprint for Human-Like Tuberculosis in Mice, 21st Century Pathology, Volume 6 (1): 161
Abstract
Murine models of tuberculosis have been criticized as poor surrogates for human disease because standard inbred strains rarely develop the necrotic, hypoxic granulomas that dominate advanced human pulmonary tuberculosis. Guiewi Makafe et al. show that this limitation reflects model selection rather than an intrinsic failure of the mouse. In Nos2‑deficient mice, they establish aerosol-route infection and a vaccine‑primed challenge system (immunization with mc²6230, H37Rv ΔRD1 ΔpanCD, followed by virulent Mtb challenge) that yields reproducible hypoxic necrotic lesions whose microenvironment contributes to poor chemotherapy response. This work strengthens the case for pathology‑relevant murine platforms while providing a timely opportunity to correct a persistent misconception: reactive nitrogen and oxygen species (RNS/ROS) are essential regulators of inflammation and lesion evolution in tuberculosis, but they should not be treated as the principal sterilizing effector mechanisms in vivo. Pairing lesion‑realistic models with genetic systems that directly report immune sterilization may be the most efficient route to uncovering the macrophage execution pathways that actually kill Mtb.
Keywords:
Tuberculosis; Granuloma; Hypoxia; Nitric oxide; Reactive oxygen species; Mouse model
Description
Murine models of tuberculosis have long been criticized for failing to reproduce the necrotic, hypoxic granulomas that dominate advanced human disease. In the study highlighted here, Weinrick and colleagues [1] present a decisive advance: a Nos2?/? vaccination–challenge model that reproducibly generates hypoxic, necrotizing lung lesions following aerosol infection. This work demonstrates that varied pathology in mice is not elusive, but conditional, emerging when specific immune regulatory pathways are removed (Figure 1).
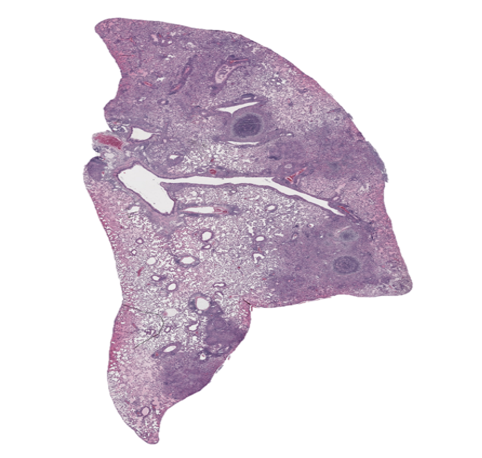
Figure 1: Human-like necrotizing tuberculosis lesions in Nos2?/? mice. Representative lung section from a Nos2?/? mouse immunized with an auxotrophic Mycobacterium tuberculosis strain and challenged with virulent Mtb, showing organized necrotizing granulomas that closely resemble the caseating pathology of advanced human tuberculosis. The image illustrates how loss of nitric oxide–mediated immune regulation reveals a form of tuberculosis pathology long thought to be absent in mice, reframing necrosis as a consequence of unrestrained immune pressure rather than failed host defense. Image courtesy of Brian Weinrick.
The conceptual foundation for this advance was laid by Igor Kramnik, whose genetic dissection of host susceptibility by mapping the sst1 locus demonstrated that tuberculosis pathology is governed by host immune set-points rather than bacterial burden alone [2,3]. Kramnik’s C3HeB/FeJ model decisively overturned the notion that mice are intrinsically incapable of modelling human tuberculosis, reframing pathology as an active, genetically controlled outcome of immune pressure. Subsequent studies of “Collaborative Cross” mice by Sassetti and colleagues revealed the contribution of a variety of loci to infection outcomes [4]. Weinrick’s Nos2?/? model extends this lineage by identifying nitric oxide as a central regulator of lesion architecture, rather than as a dominant sterilizing effector.
This conclusion resonates strongly with Barry Bloom’s enduring observation that “pathology is the price you pay for protection” [5]. In Weinrick’s system, loss of nitric oxide removes a decisive governor, allowing immune pressure to proceed at the cost of necrosis, hypoxia, and impaired drug penetration—features that closely mirror advanced human tuberculosis. The resulting lesions are not evidence of failed immunity, but of unrestrained protection.
Decades of work have established the biological importance of reactive nitrogen intermediates. Seminal studies by Carl Nathan defined inducible nitric oxide synthase (iNOS) as a powerful antimicrobial system and a major protective locus in murine tuberculosis [6,7]. While work from John Chan and colleagues demonstrated that reactive nitrogen intermediates exert potent antimycobacterial pressure in vitro and contribute to immune control in vivo [8]. However, these same studies also revealed a critical limitation: despite sustained iNOS expression, Mycobacterium tuberculosis commonly persists, indicating that nitric oxide functions predominantly as a bacteriostatic and immunoregulatory force, rather than as a consistent execution mechanism [9].
Human immunology provides an important complementary perspective. Robert Modlin and colleagues demonstrated that cytolytic T cells can directly reduce M. tuberculosis viability through a granule-dependent mechanism mediated by granulysin, bypassing the action of nitric oxide in the phagocyte altogether [10]. At the microbial level, Heran Darwin and colleagues showed that M. tuberculosis devotes substantial genetic resources—including its proteasome—to resisting nitrosative stress, underscoring both the potency of nitric oxide and the evolutionary reality that it is rarely decisive [11].
Together, these lines of evidence sharpen the conceptual impact of Weinrick’s work. The Nos2?/? model does not reveal nitric oxide–dependent sterilization; instead, it clarifies how immune regulation shapes pathology. This distinction between immune control and immune killing has often been blurred. Weinrick’s contribution is to separate them experimentally.
Becker and colleagues further sharpens this distinction by demonstrating that CD4? T cells control Mycobacterium tuberculosis through cognate MHCII-dependent interactions with monocyte-derived macrophages that induce a glycolytic metabolic program, rather than relying solely on interferon-γ–driven nitric oxide production [12]. In their model, IFN-γ is essential for recruitment of infectable macrophages, yet bacterial control requires direct T cell recognition and metabolic reprogramming of infected cells. These findings reinforce the view that nitric oxide functions primarily as a regulator of inflammation and bacterial physiology, while adaptive immunity may deploy distinct, non-classical effector mechanisms to achieve control.
In this context, the adaptive immune sterilization of the conditionally persistent strain mc²7901 provides a crucial counterpoint. In immunocompetent mice, mc²7901 is eliminated, whereas in Rag1?/? mice lacking adaptive immunity it persists for more than a year, demonstrating unequivocally that adaptive immunity can sterilize M. tuberculosis under defined conditions [13]. Forthcoming work indicates the sterilization of mc²7901 occurs in the absence of NOS2 and Phox91, consistent with emerging evidence that CD4? T cell–mediated metabolic programming of infected macrophages may constitute a nitric oxide–independent pathway of mycobacterial control [12].
Taken together, the work of Kramnik, Bloom, Chan, Nathan, Sasetti, Modlin, Darwin, and now Weinrick forms a coherent trajectory rather than a contradiction. Nitric oxide emerges not as an executioner but as a judge, a critical regulator of inflammation, pathology, and bacterial physiology. Weinrick’s Nos2?/? model shows us what protection looks like when that regulation is removed. The mc²7901 system shows us that sterilization is possible—and that its mechanism remains to be discovered.
Acknowledgments
This work is supported by grants from the National Institutes of Health (AI 26170, R24AI134650 R01AI192152).
Conflicts of Interest
The author declares no conflicts of interest.
Author Contributions (ICMJE)
Conceptualization, drafting, and final approval: WRJ.
IRB Approval
Not applicable (no human subjects research).
References
- Makafe GG, Low-Beer T, Travis K, Cole L, Bernacki D, Duso D, et al. Mouse models uniformly featuring human-like lesions harboring drug-tolerant Mycobacterium tuberculosis. bioRxiv. 2025. https://www.biorxiv.org/content/10.1101/2025.
- Apt A, Kramnik I. Man and mouse TB: contradictions and solutions. Tuberculosis (Edinb). 2009;89(3):195-8. https://doi.org/10.1016/j.tube.2008.12.002
- Driver ER, Ryan GJ, Hoff DR, Irwin SM, Basaraba RJ, Kramnik I, et al. Evaluation of a mouse model of necrotic granuloma formation using C3HeB/FeJ mice for testing of drugs against Mycobacterium tuberculosis. Antimicrob Agents Chemother. 2012;56(6):3181-95. https://doi.org/10.1128/AAC.06279-11
- Smith CM, Proulx MK, Olive AJ, Laddy D, Mishra BB, Moss C, et al. Tuberculosis susceptibility and vaccine protection are independently controlled by host genotype. mBio. 2016;7(5). https://doi.org/10.1128/mBio.01516-16
- W GA, Bloom BR. Mechanisms of pathogenesis in tuberculosis. In: Bloom BR, editor. Tuberculosis: Pathogenesis, Protection and Control. American Society for Microbiology. 1994;485-501. https://doi.org/10.1128/9781555818357.ch28
- Nathan C, Shiloh MU. Reactive oxygen and nitrogen intermediates in the relationship between mammalian hosts and microbial pathogens. Proc Natl Acad Sci U S A. 2000;97(16):8841-8. https://doi.org/10.1073/pnas.97.16.8841
- MacMicking JD, North RJ, LaCourse R, Mudgett JS, Shah SK, Nathan CF. Identification of nitric oxide synthase as a protective locus against tuberculosis. Proc Natl Acad Sci U S A. 1997;94(10):5243-8. https://doi.org/10.1073/pnas.94.10.5243
- Chan J, Xing Y, Magliozzo RS, Bloom BR. Killing of virulent Mycobacterium tuberculosis by reactive nitrogen intermediates produced by activated murine macrophages. J Exp Med. 1992;175(4):1111-22. https://doi.org/10.1084/jem.175.4.1111
- Flynn JL, Scanga CA, Tanaka KE, Chan J. Effects of aminoguanidine on latent murine tuberculosis. J Immunol. 1998;160(4):1796-803. https://www.jimmunol.org/content/160/4/1796
- Stenger S, Hanson DA, Teitelbaum R, Dewan P, Niazi KR, Froelich CJ, et al. An antimicrobial activity of cytolytic T cells mediated by granulysin. Science. 1998;282(5386):121-5. https://doi.org/10.1126/science.282.5386.121
- Darwin KH, Ehrt S, Gutierrez-Ramos JC, Weich N, Nathan CF. The proteasome of Mycobacterium tuberculosis is required for resistance to nitric oxide. Science. 2003;302(5652):1963-6. https://doi.org/10.1126/science.1091493
- Becker SH, Ronayne CE, Bold TD, Jenkins MK. Antigen-specific CD4+ T cells promote monocyte recruitment and differentiation into glycolytic lung macrophages to control Mycobacterium tuberculosis. PLoS pathog.2025;21(6),e1013208. https://doi.org/10.1371/journal.ppat.1013208
- Vilchèze C, Porcelli SA, Chan J, Jacobs Jr WR. Sterilization by adaptive immunity of a conditionally persistent mutant of Mycobacterium tuberculosis. mBio. 2021;12(1):10-128. https://doi.org/10.1128/mBio.02735-201.