Clinicopathological Correlates of COVID-19 Pandemic Waves and Long COVID
Authors: L. Maximilian Buja*, Michelle M. McDonald, Bihong Zhao
Department of Pathology and Laboratory Medicine, McGovern Medical School, The University of Texas Health Science Center at Houston (UTHealth), Houston, Texas, USA
*Correspondence to: Professor. L. Maximilian Buja, Department of Pathology and Laboratory Medicine, McGovern Medical School, The University of Texas Health Science Center at Houston (UTHealth), Houston, Texas, USA; Email: l.maximilian.buja@uth.tmc.edu
Received: May 03, 2022; Accepted: June 06, 2021; Published: June 14, 2021
Citation: Buja LM, McDonald MM and Zhao B (2022) Clinicopathological Correlates of COVID-19 Pandemic Waves and Long COVID, 21st Century Pathology, Volume 2 (3): 122
Abstract
After the onset of the Coronavirus Disease 2019 (COVID-19) pandemic, there have been four waves of infection related to the spread of the causative virus, SARS-CoV-2, and its mutant variants. Also, a condition of post-acute sequelae of SARS-CoV-2 (PASC) or long-haul/long COVID-19 has been recognized. In this report, we update the clinicopathological findings related to the four COVID-19 waves and long COVID-19.
Keywords:
SARS-CoV-2; COVID-19 Waves; Long Covid-19; Troponin; D-dimer; Coagulopathy; Heart; Lung
Highlights
- Ongoing mutation of the initial SRS-CoV-2 virus has produced mutant strains linked to four waves of COVID-19.
- Initial autopsy studies have documented that COVID-19 produces a distinctive form of diffuse alveolar damage (DAD) or a variant acute fibrinous and organizing pneumonia (AFOP) with systemic manifestations associated with thrombotic microangiopathy.
- The incidence of lymphocytic myocarditis in COVID-19 is less than 5% whereas a subset of cases exhibits a distinctive macrophage-dominant inflammatory process which also may be seen in some cases of advanced heart failure as a previously unrecognized form of inflammatory heart disease.
- Although each wave has distinctive epidemiological features and one study found less cardiac inflammation and micro thrombosis in second wave cases, major differences in pathology of fatal cases have not been manifest based on our experience with 83 autopsy cases; more autopsy studies are needed to address this issue.
- Post-acute sequelae of SARS-CoV-2 (PASC) or long-haul/long COVID-19 affect significant numbers of survivors of acute COVID-19; The NIH is embarking on a major effort to obtain definitive information regarding the clinical and pathological features of this condition.
Introduction
Shortly after the onset of the Coronavirus Disease 2019 (COVID-19) pandemic, the causative agent was identified as the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus [1]. There have been several factors which have influenced the course of the pandemic and the response of the medical and public health communities. These are 1) Documentation of the myriad of clinical manifestations of the disease [1-9]; 2) Elucidation of the basic biology of viral infectivity [10-15]; 3) Implementation of diagnostic testing and various public health containment measures [16]; 4) Autopsy studies which have defined the basic pathology of the disease [17-22] and have provided the factual basis for models of pathogenesis [23-25]; 5) Pathology studies which have clarified the nature of cardiovascular system involvement [26-35]; 6) Ongoing mutation of the virus resulting in variants with modified biological effects [11, 36-41]; 7) Development and administration of safe and effective vaccines [10, 36-39]; and 8) Development of multimodal treatment protocols influenced by clinical and autopsy pathology findings [42-48]. Nevertheless, in two and a half years, the COVID-19 pandemic has resulted in over five hundred million confirmed cases and over six million deaths reported by the World Health Organization (WHO) (https://covid19.who.int/). The purpose of this review is to provide an update on the current understanding of this impactful disease based on the correlation of clinical and pathological observations and studies with a perspective on the ongoing evolution of the pandemic.
Virus-Host Interaction
SARS-CoV-2 infection is initiated by binding of the spike protein (S) of the SARS-CoV-2 virus to a complex of the ACE-2 receptor and accessory proteases, the transmembrane serine protease 2 receptor (TMPRSS2) and cathepsin L (CTSL), expressed on respiratory epithelial cells in the upper respiratory tract and on type II pneumocytes, endothelial cells and alveolar macrophages in the lungs [1-4, 10]. The distribution of the ACE-2 receptor with the accessory proteases in various cell types in different organs is a prime determinant of the involvement of these organs in COVID-19. Molecular biological studies have provided the best evidence regarding cell types infected by the virus and the persistence of viral infectivity [10-15].
Clinical Features of Acute and Chronic Illness
ACOVID-19 presents as an acute, febrile, respiratory illness [1-4]. Although many individuals infected with SARS-CoV-2 may be asymptomatic or have mild flu-like symptoms, the respiratory condition can progress to acute respiratory distress syndrome (ARDS), and some individuals can develop a severe illness with multiorgan involvement and severe hyper inflammation [1-9]. Paradoxically, cytokine levels in the blood, including interleukin-6 (IL-6), are moderately elevated and less than levels seen in sepsis [8, 9]. Prognostic indicators of a more serious and potentially fatal course include older age, lymphopenia, elevated D-dimer level, elevated troponin levels, and the co-morbidities of pre-existing cardiovascular disease, hypertension, obesity, diabetes mellitus, and renal disease. Clinical evidence of cardiovascular involvement portents an adverse, and often fatal, outcome [1-7]. While many subjects have an early recovery, some have persistent systemic and neurological symptoms as manifestations of a condition with the formal designation of post-acute sequelae of SARS-CoV-2 (PASC) also known as long-haul or long COVID-19 [49, 50].
Clinicopathological Correlates of COVID-19 Virus Variants and Pandemic Waves
The COVID-19 pandemic was initiated in late 2019 and early 2020 apparently due to a new strain of the SARS-CoV-2 virus which was capable of transmission from animal vectors to man [1-4]. This strain was responsible for the initial spread and wave of COVID-19. Since then, mutations of the initial virus have given rise to successive variants of concern (VOC) which have produced waves and peaks of COVID-19 around the world (Table 1) [11, 36-41]. To date, the incidence of cases has waxed and waned with four peak periods or waves that have been linked to the spreading of the initial virus followed by the spread of mutant strains [11, 36-41].
|
New WHO name |
Strain name |
Other name(s) |
Major mutation sites |
First reported place |
First reported time |
Therapeutics |
|
Alpha |
D614G |
VOC-202012/01, or 201/501Y.V1 |
D614G |
German, China |
Jan-20 |
Pfizer-BioNTech vaccine showed decreased neutralization; Moderna no changes. |
|
B.1.1.7 |
D614G, a two amino acid deletion at positions 69-70, N501Y, P681H |
UK |
Dec-20 |
Moderna and Novavax vaccines showed decreased neutralization. |
||
|
B.1.526 |
E484K, S447N, L5F, T95I, D253G, D614G, A701V |
New York, USA |
Nov-20 |
Pfizer-BioNTech and Moderna vaccines showed no change in S477N, but decreased neutralization on E484K. |
||
|
Beta |
B.1.351 |
501Y.V2 |
K417N, E484K, N501Y |
South Africa |
Late 2020 |
NVX-CoV2373 vaccine showed good neutralization; Moderna vaccine showed decreased neutralization. |
|
B.1.1.28.1 |
E484K |
Brazil |
Feb-20 |
Covaxin vaccine showed good neutralization; Pfizer-BioNtech and Moderna vaccines showed decreased neutralization. |
||
|
Gamma |
P1 |
20J/501Y.V3, belongs to B.1.1.28 strain lineage |
K417T, E484K, N501Y |
Brazil, Japan |
Early 2021 |
mAb effects vary; Pfizer-BioNtech and Moderna vaccines showed decreased neutralization. |
|
Delta |
B.1.617 |
D111D, G142D, L452R, E484R, E484Q, D614G, P681R |
India |
Late 2020 |
Pfizer-BioNtech and Moderna vaccines showed decreased neutralization. |
|
|
Omicron |
B.1.1.529* |
30 in S-protein, N501Y, Q498R, H655Y,inclN679K,P681H |
South Africa |
Late 2021 |
Major vaccines showed decreased neutralization |
|
|
*Omicron Subvariants BA.1 (B.1.1.529.1), BA.2 (B.1.1.529.2), and BA.3 (B.1.1.529.3). Adapted with permission from Forchette L, Sebastian W, Liu T. A comprehensive review of COVID-19 virology, vaccines, variants, and therapeutics. Curr Med Sci. 2021;41(6):1037-1051. doi: 10.1007/s11596-021-2395-1. |
||||||
In 2020, the first two waves of the pandemic occurred in many countries around the world, including Western Europe: the first between February/March and the end of May/June, and the second from September until the end of the year. Differences have been reported in the disease course and mortality between first and second wave COVID-19 patients, including differences in patient demographics, disease course, and mortality between first and second wave COVID-19 [34, 37-39]. In 2021 and 2022, two subsequent waves of COVID-19 occurred, each with distinctive demographics and outcomes [11, 41, 42].
Since the onset of the pandemic, four waves and peaks of COVID-19 cases have occurred in Houston, Texas (Figures 1 and 2). The epidemiology and biological responses have been well documented [51-56]. At our institution, McGovern Medical School, and affiliated hospitals in Houston, obstacles were overcome to keep our autopsy service operational in the early days of the pandemic. This resulted in the publication of one of the first autopsy reports of fatal COVID-19 in the USA [19, 20]. Subsequently, there have been four peaks of fatal cases coming to autopsy at our institution corresponding to the peaks of SARS-CoV-2 viral spread in the Houston community (Figure 3). To date, we have performed 83 autopsies on patients who had a positive polymerase chain reaction (PCR) nasal swab test for the SARS-CoV-2 virus during their final hospitalization.
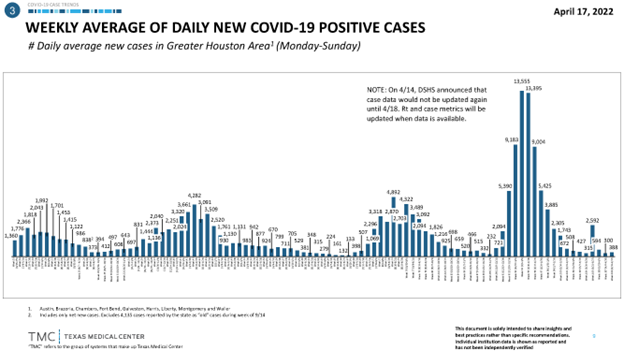
Figure 1: Weekly average of daily new COVID-19 positive cases in Houston, Texas.
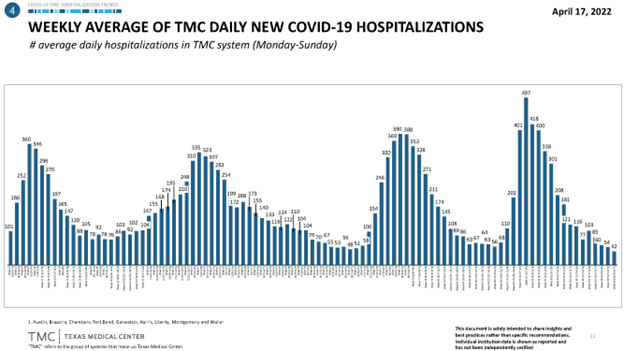
Figure 2: Weekly average of Texas Medical Center (TMA) daily new COVID-19 hospitalizations.
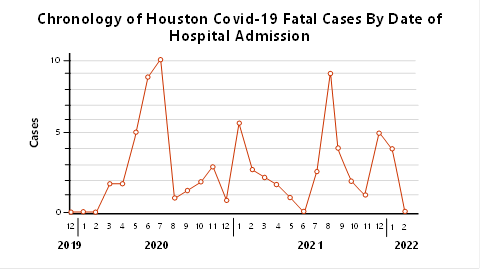
Figure 3: Chronology of autopsy cases performed on the Autopsy Service of the Department of Pathology and Laboratory Medicine from patients with fatal COVID-19 at affiliated hospitals. Note the temporal relationship of peaks to peaks in daily new COVID-19 positive cases and hospitalizations in Houston, Texas. The four peaks correspond to the prevalence in the community of the wild-type virus followed by the alpha and beta strains, then the gamma and delta strains, and most recently, the omicron strain.
We reviewed the autopsy reports and our data files pertaining to our 83 autopsy cases. We did not detect any consistent trends or differences in cases coming to autopsy related to the four waves of disease in Houston. We were not impressed with differences in cardiac inflammation, cardiomyocyte injury, and microvascular thrombogenicity over time as reported by Wu L, et al. (2022) [34]. Descriptions of the pathology of the pulmonary, cardiovascular, neurologic, and other organ systems mostly have been based on autopsy studies of fatal cases during the first COVID-19 wave. Further studies are needed to address pathological findings in fatal cases from the different waves of the pandemic. Details of the pathological studies are presented below.
Autopsy Studies and Clinicopathological Correlation
Our first study collated the pathological findings from initial published autopsy reports on 23 patients with coronavirus disease 2019 (COVID-19) from 5 centers in the United States of America, including 3 cases from Houston, Texas [19, 20]. Subsequently, we extended the analysis to include 34 of our Houston cases [21]. Our studies document that pulmonary manifestation of COVID-19 respiratory disease correlates with patients? duration of illness, having features of extensive diffuse alveolar damage (DAD), including 1) Acute/exudate phase in early (5-7 days) illness patients, with hyaline membranes, type II pneumocyte hyperplasia, low grade lymphohistiocytic infiltrate, and microvasculopathy with intravascular and extravascular ?brin deposition; 2) Proliferative/organizing phase in patients over 1-week illness, with the proliferation of fibroblast and deposition of newly synthesized collagens; and 3) Fibrotic phase in patients over 3 weeks illness with alveolar septal fibrosis, mimic a fibrotic NSIP. In the long-illness group of patients, a peculiar finding is cystic degenerative changes presented by groups of cystic spaces with dystrophic calcification in the peripheral regions, sometimes with multi-nucleated giant cells. We think the cystic change could be related to ischemic change secondary to many micro-thrombi, and/or mechanic damage by mechanic ventilation. There are overlapping features of 2 out of 3 phases in the same lungs. We have also found features of intravascular trapping of megakaryocytes, platelets and neutrophils in capillaries demonstrated by electron microscopy and microthrombi observed by light microscopy in a majority (59%) of cases. Macrothrombi in the form of pulmonary thromboemboli in segmental pulmonary arteries with pulmonary infarcts and/or hemorrhage were observed in a quarter of the cases. In half of the cases, super-imposed bacterial pneumonia was present (Table 2).
Table 2: Summary of Clinical and Pathological Manifestations of Severe Coronavirus Disease 2019 (COVID-19).
|
Systems |
Cardiovascular |
Respiratory |
CNS |
|
Clinical Features |
Heart failure symptoms ST-segment changes on electrocardiogram |
Acute respiratory distress syndrome (ARDS) Variable clinical manifestations and pathological findings |
Anosmia (loss of smell) Ageusia (loss of taste) Fatigue Sensory and/or motor defects |
|
Pathology |
Increased interstitial macrophages Focal capillary endothelilitis Focal individual cardiomyocyte necrosis microthrombi Rare lymphocytic myocarditis |
Diffuse Alveolar Damage (DAD) Exudative phase, with hyaline membranes Lymphohistocytic inflammation Pneumocyte hyperplasia and dysplasia Endothelialitis Microthrombi, macrothrombi acute fibrinous and organizing pneumonia (AFOP) (variant) Progression of DAD, proliferative and fibrotic phases
|
Thrombi Infarcts Gliosis |
|
Comorbidities |
Obesity, hypertension, diabetes mellitus, renal disease, chronic lung disease |
||
|
Laboratory Findings |
Lymphopenia, elevated D-dimer, elevated troponin |
||
|
CNS indicates Central Nervous System. Adapted with permission from Barth RF, Buja LM, Barth AL, et al. (2021) A Comparison of the Clinical, Viral, Pathologic, and Immunologic Features of Severe Acute Respiratory Syndrome (SARS), Middle East Respiratory Syndrome (MERS), and Coronavirus 2019 (COVID-19) Diseases. Arch Pathol Lab Med. 145:1194-1211. doi: 10.5858/arpa.2020-0820-SA. |
|||
Some patients exhibited a variant pattern of acute lung injury known as acute fibrinous and organizing pneumonia (AFOP) characterized by interstitial pneumonitis without hyaline membranes and with intra-alveolar deposits of fibrin enclosed in granulation tissue. Our findings have been confirmed by others [1, 17, 18, 22, 57].
The pulmonary pathology of COVID-19 shares features with influenza and other causes of acute lung injury [58, 59]. However, COVID-19 pneumonitis is distinctive for its severity and extension of parenchymal involvement and associated thrombotic microangiopathy [19-21, 23, 24].
In addition to major pulmonary pathology, our Houston cases showed subtle changes in the heart consisting of occasional small foci of acute injury of cardiomyocytes without in?ammatory cellular in?ltrates, and thrombi in intramural coronary arteries as well as focal lymphocytic epicarditis [19-21]. Large and small infarcts and hemorrhages often were present in the brains. Other changes included depletion of splenic white pulp, focal hepatocellular degeneration, and rare glomerular capillary thrombosis. Thus, the autopsy ?ndings provide definitive evidence for the concept that the pathogenesis of severe COVID-19 disease involves initial viral-induced injury of multiple organs, including the heart and lungs, coupled with an intense inflammatory reaction, microangiopathy and prothrombotic coagulopathy [23-25].
The autopsy studies provided insights into pathogenesis that contributed to the development of multimodal therapeutic protocols which have evolved over the course of the pandemic. These insights focused on the roles of persistent hyper inflammation, hypercoagulability, and microangiopathy. Current multimodal therapy includes monoclonal antibodies, corticosteroids, anticoagulation, and protocols such as MATH+ (methylprednisolone, ascorbic acid, thiamine, and heparin) [42-48].
COVID-19 and the Cardiovascular System
Our work has been part of a concerted effort by cardiovascular pathologists, individually and through their organizations, the Society for Cardiovascular Pathology (SCVP) and the Association for European Cardiovascular Pathology (AECVP), to obtain and disseminate credible information about the pathological basis for the diverse clinical manifestations of cardiovascular system involvement in COVID-19 [26]. An important issue to be addressed was to sort out the pathological basis for the elevated troponin levels and electrocardiographic changes frequently seen in hospitalized COVID-19 patients since an initial general clinical impression was that the frequently observed elevated troponin levels equated to the frequent occurrence of life-threatening myocarditis in these patients.
An international consortium of cardiovascular pathologists initially addressed the nature of cardiac involvement in COVID-19 with a comprehensive evaluation of 21 cases of fatal COVID-19. The presence of myocarditis, as defined by the presence of multiple foci of inflammation with associated myocyte injury, was determined, and the inflammatory cell composition was analyzed by immunohistochemistry [26]. Lymphocytic myocarditis was present in only three (14%) of the cases and mild pericarditis in four (19%) of the cases. However, increased interstitial macrophage infiltration was present in 18 (86%) of the cases. Acute myocyte injury in the right ventricle, most probably due to strain/overload, was also present in four (19%) of the cases. Conclusions of the study were as follows: 1) In SARS-CoV-2 there are increased interstitial macrophages in a majority of the cases and multifocal lymphocytic myocarditis in a small fraction of the cases; 2) Other forms of myocardial injury are also present in these patients, and 3) The macrophage infiltration may reflect underlying diseases rather than COVID-19, but the macrophage infiltrates were often more intense than typically caused by underlying conditions [26].
The finding of a measurable but relatively low frequency of myocarditis have been confirmed by other cardiovascular pathologists based on case series and two literature reviews of nearly 300 patients [18-20, 26-28]. The pathology studies also have documented the presence of other histopathologic findings including focal cardiomyocyte necrosis, macro- or microvascular thrombi, inflammation, and intraluminal megakaryocytes, and found that collectively these non-myocarditis changes are much more frequent than is myocarditis.
Conclusion
Thus, pathological studies have shown: 1) A low incidence (less than 5%) of lymphocytic myocarditis [26-28] and 2) The more frequent occurrence in nearly 50% of reported cases of other histopathologic findings including focal cardiomyocyte necrosis, thrombi and megakaryocytes in intramyocardial coronary vessels, and macro-phage-dominant interstitial inflammation, as correlates of the elevated troponin levels [18-20, 26-28].
The correlation of clinical and pathological findings indicates that the cardiac pathology is part of systemic cardiovascular involvement in severe COVID-19. Clinical evidence of cardiovascular involvement in COVID-19 portents an adverse, and often fatal, outcome [1-9]. Biomarkers of cardiovascular involvement in patients with COVID-19 include elevated blood levels of D-dimers and cardiac troponin. Patients with severe COVID-19 also have elevated inflammatory markers including interleukin-6 (IL-6) and C reactive protein (CRP) as well as elevations in von Willebrand factor (VWF) and P-selectin released from perturbed endothelial cells. The systemic endothelial vascular injury likely is a common trigger for both the inflammatory and thrombotic complications of COVID-19. COVID-19 is a microvascular disease [25]. COVID-19 patients can present with the thromboembolic vascular disease with thrombi which are uniquely resistant to thrombolysis, difficult to extract, and exhibit distinctive properties in vitro on thrombo-elastography (TEG) and have evidence of fibrinolysis shutdown [60-63]. The lesson learned is that rapid implementation of maximal anticoagulation of seriously ill COVID-19 patients is essential to lessen the likelihood of the development of pulmonary and systemic thromboembolic disease.
The descriptions of the pathology of the cardiovascular system as well as the pulmonary, neurologic and other organ systems were based on autopsy studies of fatal cases during the first COVID-19 wave. Wu L, et al (2022) [34] undertook a study to analyze and compare cardiac pathology between first wave (cases from March-April 2020) and second wave (cases from October-December 2020) COVID-19 patients in Europe. Autopsied hearts from first wave (n = 15) and second wave (n = 10) COVID-19 patients and from 18 non-COVID-19 control patients were analyzed by immunohistochemistry. CD45+ leukocyte, CD68+ macrophage and CD3+ T lymphocyte infiltration, cardiomyocyte necrosis and microvascular thrombosis were quantified. In addition, the procoagulant factors tissue factor (TF), Factor VII (FVII), Factor XII (FXII), the anticoagulant protein Dipeptidyl Peptidase 4 (DPP4) and the advanced glycation end-product N(?)-Carboxymethyllysine (CML), as markers of microvascular thrombogenicity and dysfunction, were quantified. Cardiac inflammation was significantly decreased in second wave compared to first wave COVID-19 patients, predominantly related to a decrease in infiltrated lymphocytes and the absence of lymphocytic myocarditis. This was accompanied by significant decreases in cardiomyocyte injury and microvascular thrombosis. Also, microvascular deposits of FVII and CML were significantly lower in second wave compared to first wave COVID-19 patients. Wu et al. concluded that in this cohort of fatal COVID-19 cases, cardiac inflammation, cardiomyocyte injury and microvascular thrombogenicity were significantly decreased in the second wave compared to the first wave of COVID-19 [34]. The findings in this relatively small case series need to be confirmed in additional studies.
Further studies have been performed to assess the occurrence and nature of myocardial inflammation in COVID-19 and other conditions. Fox SE, et al. (2021) [29] performed an analysis with high resolution whole slide image scanning and quantification of CD3, CD4, CD8 and CD68 staining cells in myocardium of 10 COVID patients (cases from March-September 2020), 5 community acquired myocarditis and 10 controls. The analysis revealed a skewed distribution of CD68 positive cells in COVID-19 hearts with upper quantiles showing a significant increase compared to both control hearts and hearts with inflammatory myocarditis. Hearts from inflammatory myocarditis contained more CD4+ and CD8+ cells compared to both COVID-19 and control hearts. Fox SE, et al. (2021) [29] concluded that the diffuse interstitial and microvascular CD68+ cell infiltrate is distinct from typical viral myocarditis and may represent a unique form of cardiac inflammation affecting at least some COVID-19 patients.
Goldman BI, et al. (2022) reported 6 patients with sudden onset of heart failure whose endomyocardial biopsies showed myocardial inflammation with infiltrates of mostly CD68- positive macrophages and markedly fewer T lymphocytes. The entity was labeled as heart failure with CD68 predominant inflammation (?histiocytic? myocardial inflammatory disease, HMID) (30). These cases of HMID were not associated with COVID-19 infection. A rare case of fulminant myocarditis with CD68 histiocyte-dominant infiltrates has been reported in association with delta variant SARS-Co-V2 infection [64].
Bearse M, et al. (2021) [33] analyzed 41 consecutive autopsies of patients with fatal COVID-19 (cases from March ? July 2020) to investigate the associations between cardiac inflammation, myocarditis, cardiac infection by SARS-CoV-2, clinical features, laboratory measurements, and treatments. Cardiac infection was assessed by in situ hybridization and NanoString transcriptomic profiling. Cardiac infection by SARS-CoV-2 was present in 30/41 cases: virus positive with myocarditis (n = 4), virus positive without myocarditis (n = 26), and virus negative without myocarditis (n = 11). In the cases with cardiac infection, SARS-CoV-2 positive cells in the myocardium were rare, with a median density of 1 cell/cm2. Virus positive cases showed higher densities of myocardial CD68+ macrophages and CD3+ lymphocytes, as well as more electrocardiographic changes (23/27 vs 4/10; p = 0.01). Myocarditis was more prevalent with IL-6 blockade than with non-biologic immunosuppression, primarily glucocorticoids (2/3 vs. 0/14; p = 0.02). Overall, SARS-CoV-2 cardiac infection was less prevalent in patients treated with non-biologic immunosuppression (7/14 vs. 21/24; p = 0.02). Myocardial macrophage and lymphocyte densities overall were positively correlated with the duration of symptoms but not with underlying comorbidities. Bearse M, et al. (2021) [33] concluded that: 1) Cardiac infection with SARS-CoV-2 is common among patients dying from COVID-19 but with a very low number of infected cells; 2) Cardiac infection by SARS-CoV-2 is associated with more cardiac inflammation and electrocardiographic changes, and 3) Non-biologic immunosuppression is associated with lower incidences of myocarditis and cardiac infection by SARS-CoV-2.
The low incidence of lymphocytic myocarditis observed in autopsy studies has been confirmed in a study of 112 hospitalized patients [35]. A total of 112 patients with suspected acute myocarditis (AM) from 56,963 hospitalized patients with COVID-19 were evaluated between February 1, 2020, and April 30, 2021. Inclusion criteria were based on clinical, laboratory, and imaging findings. Acute myocarditis prevalence among hospitalized patients with COVID-19 was 2.4 per 1000 hospitalizations based on a definite/probable diagnosis with endomyocardial biopsy confirmation in some cases and 4.1 per 1000 including cases in the possible as well as definite/probable categories [35]. The acute myocarditis incidence of 2?4% in this clinical study confirms a similar incidence documented in autopsy studies (18-20, 26-28). A low incidence of lymphocytic myocarditis also has been documented following the administration of anti-COVID-19 mRNA vaccines, with a predilection for young men [65-69]. Most patients have a short illness and recovery, but a few fatal cases have been reported [65]. With endomyocardial biopsy or autopsy, the myocarditis has a mixed inflammatory cellular infiltrate including eosinophils [65]. The benefits of vaccination far exceed the risk of developing vaccine-related myocarditis [69].
Based on an analysis of recent studies, Fox SE, et al. (2021) and Vander Heide (2022) [29, 31, 32] proposed the following: 1) The findings of significantly increased numbers of cardiac macrophages in a relevant subset of patients with SARS-CoV-2 infection may represent a different and perhaps unique form of cardiac inflammation in patients with severe acute COVID-19; 2) The preponderance of CD68 positive cells in COVID-19 hearts and in some patients with heart failure highlight the current limitations of the definition of myocarditis, especially when applied to autopsy specimens, and 3) Given the emergence of long COVID and the unknown long-term consequences of mild to moderate COVID-19 infections, ongoing investigation of the role of macrophage accumulation in the myocardium is warranted.
PASC and Long-Haul COVID
The world continues to contend with successive waves of COVID-19 fueled by the emergence of viral variants. At the same time, persistent, prolonged, and often debilitating sequelae are increasingly recognized in convalescent individuals. The symptom complex has been designated as post-acute sequelae of SARS-CoV-2 (PASC) and also is known as 'post-COVID-19 syndrome' or 'long-haul COVID' [49, 50, 70-76]. Clinical symptomatology includes fatigue, malaise, dyspnea, defects in memory and concentration, and a variety of neuropsychiatric syndromes as the major manifestations, and several organ systems can be involved. The underlying pathophysiological mechanisms are poorly understood at present.
Many patients with mild or severe COVID-19 do not make a full recovery and have a wide range of chronic symptoms for weeks or months after infection, often of a neurological, cognitive, or psychiatric nature. PASC syndrome is defined by persistent clinical signs and symptoms that appear while or after suffering COVID-19 and persist for more than 12 weeks, often with fluctuations and relapses. The condition may overlap with or be complicated by post-viral chronic fatigue syndrome, sequelae of infection in multiple organs, and the effects of severe hospitalization/post-intensive care syndrome. PASC has been reported in patients with mild as well as severe COVID-19 and irrespective of the severity of the symptoms in the acute phase. PACS has been reported in between 10% and 65% of survivors who had mild/moderate COVID-19.
An abnormal or excessive autoimmune and inflammatory response has been postulated to play an important role in the neurological manifestations of COVID-19, but the underlying mechanisms aren't well understood [1]. The development of animal models that recapitulate the neuropathological findings of autopsied brain tissue from patients who died from severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection is an important approach for elucidating the neuropathogenesis of infection and disease. Rutkai I, et al. (2022) [77] developed such a model in SARS-CoV-2 infected non-human primates with features of neuroinflammation, microhemorrhages, brain hypoxia, and neuropathology that is consistent with hypoxic-ischemic brain injury, including evidence of neuron degeneration and apoptosis. Importantly, this is seen among infected animals that do not develop severe respiratory disease, which may provide insight into neurological symptoms associated with "long COVID". The sparse virus was detected in brain endothelial cells but did not correlate with the severity of central nervous system (CNS) injury. Rutkai et al. [77] demonstrated that SARS-CoV-2 infected non-human primates are a relevant animal model for investigating COVID-19 neuropathogenesis among human subjects and proposed that their findings can advance current understanding of the neuropathogenesis of SARS-CoV-2 infection.
Conclusion
Nevertheless, systematic autopsy studies of patients dying with a recent or previous diagnosis of PASC are needed to confirm the pathological substrate in man but have not been reported to date. The NIH has launched a billion-dollar initiative to study long COVID, which includes a provision for biobanking brain specimens [78]. The logistics of obtaining such cases with appropriate clinical documentation is daunting, but the effort is essential to further understanding of long COVID.
Authors Contribution
McGovern autopsy case analysis: LMB, MMcD, BZ; Literature review: LMB; Writing ? original draft: LMB; Writing ? review and editing: MMcD and BZ
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
Running Title: Insights into COVID-19 Waves and Long COVID
The author have no conflict of interest.
Acknowledgment
There was no external funding support for this study.
References
1. Barth RF, Buja LM, Barth AL, Carpenter DE, Parwani AV. A Comparison of the clinical, viral, pathologic, and immunologic features of severe acute respiratory syndrome (SARS), middle east respiratory syndrome (MERS), and coronavirus 2019 (COVID-19) diseases. Archives of Pathology & Laboratory Medicine. 2021 Oct;145(10):1194-211. https://doi.org/10.5858/arpa.2020-0820-SA
2. Geng YJ, Wei ZY, Qian HY, Huang J, Lodato R, Castriotta RJ. Pathophysiological characteristics and therapeutic approaches for pulmonary injury and cardiovascular complications of coronavirus disease 2019. Cardiovascular Pathology. 2020 Jul 1;47:107228. https://doi.org/10.1016/j.carpath.2020.107228
3. Madjid M, Safavi-Naeini P, Solomon SD, Vardeny O. Potential effects of coronaviruses on the cardiovascular system: a review. JAMA cardiology. 2020 Jul 1;5(7):831-40. https://doi.org/10.1001/jamacardio.2020.1286
4. Wiersinga WJ, Rhodes A, Cheng AC, Peacock SJ, Prescott HC. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): a review. Jama. 2020 Aug 25;324(8):782-93. https://doi.org/10.1001/jama.2020.12839
5. Nishiga M, Wang DW, Han Y, Lewis DB, Wu JC. COVID-19 and cardiovascular disease: from basic mechanisms to clinical perspectives. Nature Reviews Cardiology. 2020 Sep;17(9):543-58. https://doi.org/10.1038/s41569-020-0413-9
6. Azevedo RB, Botelho BG, Hollanda JV, Ferreira LV, Junqueira de Andrade LZ, Oei SS, Mello TD, Muxfeldt ES. Covid-19 and the cardiovascular system: a comprehensive review. Journal of human hypertension. 2021 Jan;35(1):4-11. https://doi.org/10.1038/s41371-020-0387-4
7. Salabei JK, Asnake ZT, Ismail ZH, Charles K, Stanger GT, Abdullahi AH, Abraham AT, Okonoboh P. COVID-19 and the Cardiovascular System: An Update. The American Journal of the Medical Sciences. 2022 Feb 11. https://doi.org/10.1016/j.amjms.2022.01.022
8. Liu Y, Zhang C, Huang F, Yang Y, Wang F, Yuan J, Zhang Z, Qin Y, Li X, Zhao D, Li S. Elevated plasma levels of selective cytokines in COVID-19 patients reflect viral load and lung injury. National Science Review. 2020 Jun 1;7(6):1003-11. https://doi.org/10.1093/nsr/nwaa037
9. Stolarski AE, Kim J, Zhang Q, Remick DG. Cytokine Drizzle—the rationale for abandoning “Cytokine Storm”. Shock. 2021 Nov 1;56(5):667-72. https://doi.org/10.1097/SHK.0000000000001769
10. Marian AJ. Current state of vaccine development and targeted therapies for COVID-19: impact of basic science discoveries. Cardiovascular Pathology. 2021 Jan 1;50:107278. https://doi.org/10.1016/j.carpath.2020.107278
11. Scudellari M. How the coronavirus infects cells-and why Delta is so dangerous. Nature. 2021 Jul 1;595(7869):640-4. https://doi.org/10.1038/d41586-021-02039-y
12. Desai N, Neyaz A, Szabolcs A, Shih AR, Chen JH, Thapar V, Nieman LT, Solovyov A, Mehta A, Lieb DJ, Kulkarni AS. Temporal and spatial heterogeneity of host response to SARS-CoV-2 pulmonary infection. Nature communications. 2020 Dec 9;11(1):1-5. https://doi.org/10.1038/s41467-020-20139-7
13. Massoth LR, Desai N, Szabolcs A, Harris CK, Neyaz A, Crotty R, Chebib I, Rivera MN, Sholl LM, Stone JR, Ting DT. Comparison of RNA in situ hybridization and immunohistochemistry techniques for the detection and localization of SARS-CoV-2 in human tissues. The American journal of surgical pathology. 2021 Jan 6;45(1):14-24. https://doi.org/10.1097/PAS.0000000000001563
14. Delorey TM, Ziegler CG, Heimberg G, Normand R, Yang Y, Segerstolpe Å, Abbondanza D, Fleming SJ, Subramanian A, Montoro DT, Jagadeesh KA. COVID-19 tissue atlases reveal SARS-CoV-2 pathology and cellular targets. Nature. 2021 Jul;595(7865):107-13. https://doi.org/10.1038/s41586-021-03570-8
15. Chertow D, Stein S, Ramelli S, et al. (2021) SARS-COV-2 infection and persistence throughout the human body and brain. Research Square. https://doi.org/10.21203/rs.3.rs-1139035/v1
16. To KK, Sridhar S, Chiu KH, Hung DL, Li X, Hung IF, Tam AR, Chung TW, Chan JF, Zhang AJ, Cheng VC. Lessons learned 1 year after SARS-CoV-2 emergence leading to COVID-19 pandemic. Emerging Microbes & Infections. 2021 Jan 1;10(1):507-35. https://doi.org/10.1080/22221751.2021.1898291
17. Barth RF, Buja LM, Parwani AV. The spectrum of pathological findings in coronavirus disease (COVID-19) and the pathogenesis of SARS-CoV-2. Diagnostic Pathology. 2020 Dec;15(1):1-4. https://doi.org/10.1186/s13000-020-00999-9
18. Fox SE, Akmatbekov A, Harbert JL, Li G, Brown JQ, Vander Heide RS. Pulmonary and cardiac pathology in African American patients with COVID-19: An autopsy series from New Orleans. The Lancet Respiratory Medicine. 2020 Jul 1;8(7):681-6. https://doi.org/10.1016/S2213-2600(20)30243-5
19. Buja LM, Wolf DA, Zhao B, Akkanti B, McDonald M, Lelenwa L, Reilly N, Ottaviani G, Elghetany MT, Trujillo DO, Aisenberg GM. The emerging spectrum of cardiopulmonary pathology of the coronavirus disease 2019 (COVID-19): report of 3 autopsies from Houston, Texas, and review of autopsy findings from other United States cities. Cardiovascular Pathology. 2020 Sep 1;48:107233. https://doi.org/10.1016/j.carpath.2020.107233
20. Buja LM, Zhao B, McDonald M, Ottaviani G, Wolf DA. Cardiovasc Pathol. 53:107339. https://doi.org/10.1016/j.carpath.2021.107339
21. Buja LM, McDonald M, Zhao B. COVID-19 cardiac injury: An important cause of COVID-19 related morbidity and mortality. 21st Century Pathology. 2021 Nov 1: 104.
22. Hooper JE, Padera RF, Dolhnikoff M, da Silva LF, Duarte-Neto AN, Kapp ME, Lacy JM, Mauad T, Saldiva PH, Rapkiewicz AV, Wolf DA. A Postmortem Portrait of the Coronavirus Disease 2019 (COVID-19) Pandemic: A large multi-institutional autopsy survey study. Archives of Pathology & Laboratory Medicine. 2021 May 1;145(5):529-35. https://doi.org/10.5858/arpa.2020-0786-SA
23. Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T, Laenger F, Vanstapel A, Werlein C, Stark H, Tzankov A, Li WW. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. New England Journal of Medicine. 2020 Jul 9;383(2):120-8. https://doi.org/10.1056/NEJMoa2015432
24. Gustine JN, Jones D. Immunopathology of Hyperinflammation in COVID-19. The American Journal of Pathology. 2021 Jan 1;191(1):4-17. https://doi.org/10.1016/j.ajpath.2020.08.009
25. Lowenstein CJ, Solomon SD. Severe COVID-19 is a microvascular disease. Circulation. 2020 Oct 27;142(17):1609-11. https://doi.org/10.1161/CIRCULATIONAHA.120.050354
26. Basso C, Leone O, Rizzo S, De Gaspari M, Van Der Wal AC, Aubry MC, Bois MC, Lin PT, Maleszewski JJ, Stone JR. Pathological features of COVID-19-associated myocardial injury: a multicentre cardiovascular pathology study. European Heart Journal. 2020 Oct 14;41(39):3827-35. https://doi.org/10.1093/eurheartj/ehaa664
27. Halushka MK, Vander Heide RS. Myocarditis is rare in COVID-19 autopsies: cardiovascular findings across 277 postmortem examinations. Cardiovascular Pathology. 2021 Jan 1;50:107300. https://doi.org/10.1016/j.carpath.2020.107300
28. Buja LM, Stone JR. A novel coronavirus meets the cardiovascular system: Society for cardiovascular pathology symposium 2021. Cardiovascular Pathology. 2021 Jul;53:107336. https://doi.org/10.1016/j.carpath.2021.107336
29. Buja LM, Stone JR. A novel coronavirus meets the cardiovascular system: Society for cardiovascular pathology symposium 2021. Cardiovascular Pathology. 2021 Jul;53:107336. https://doi.org/10.1016/j.carpath.2021.107361
30. Goldman BI, Choung HY, Sainvil M, Miller CW. The spectrum of macrophage-predominant inflammatory myocardial disease presenting as fulminant heart failure. Cardiovascular Pathology. 2022 Mar 1;57:107393. https://doi.org/10.1016/j.carpath.2021.107393
31. Vander Heide RS (2022) Commentary on the role of cardiac inflammation in pathology of COVID-19. Cardiovasc Pathol In press.
32. Vander Heide RS. Diffuse mononuclear inflammatory response to COVID-19: friendly fire or smoldering enemy?. Cardiovascular Pathology. 2022 May;58:107416. https://doi.org/10.1016/j.carpath.2022.107416
33. Bearse M, Hung YP, Krauson AJ, Bonanno L, Boyraz B, Harris CK, Helland TL, Hilburn CF, Hutchison B, Jobbagy S, Marshall MS. Factors associated with myocardial SARS-CoV-2 infection, myocarditis, and cardiac inflammation in patients with COVID-19. Modern Pathology. 2021 Jul;34(7):1345-57. https://doi.org/10.1038/s41379-021-00790-1
34. Wu L, Baylan U, van der Leeden B, Schurink B, Roos E, Schalkwijk CG, Bugiani M, van der Valk P, van Rossum AC, Zeerleder SS, Heunks LM. Cardiac inflammation and microvascular procoagulant changes are decreased in second wave compared to first wave deceased COVID-19 patients. International Journal of Cardiology. 2022 Feb 15;349:157-65. https://doi.org/10.1016/j.ijcard.2021.11.079
35. Ammirati E, Lupi L, Palazzini M, Hendren NS, Grodin JL, Cannistraci CV, Schmidt M, Hekimian G, Peretto G, Bochaton T, Hayek A. Prevalence, characteristics, and outcomes of COVID-19–associated acute myocarditis. Circulation. 2022 Apr 12;145(15):1123-39. https://doi.org/10.1161/CIRCULATIONAHA.121.056817
36. Forchette L, Sebastian W, Liu T. A comprehensive review of COVID-19 virology, vaccines, variants, and therapeutics. Current Medical Science. 2021 Dec;41(6):1037-51. https://doi.org/10.1007/s11596-021-2395-1
37. Fernandes Q, Inchakalody VP, Merhi M, Mestiri S, Taib N, Moustafa Abo El-Ella D, Bedhiafi T, Raza A, Al-Zaidan L, Mohsen MO, Yousuf Al-Nesf MA. Emerging COVID-19 variants and their impact on SARS-CoV-2 diagnosis, therapeutics and vaccines. Annals of Medicine. 2022 Dec 31;54(1):524-40. https://doi.org/10.1080/07853890.2022.2031274
38. Francis AI, Ghany S, Gilkes T, Umakanthan S. Review of COVID-19 vaccine subtypes, efficacy and geographical distributions. Postgraduate Medical Journal. 2022 May 1;98(1159):389-94. https://doi.org/10.1136/postgradmedj-2021-140654
39. Lin L, Zhao Y, Chen B, He D. Multiple COVID-19 waves and vaccination effectiveness in the United States. International Journal of Environmental Research and Public Health. 2022 Feb 17;19(4):2282. https://doi.org/10.3390/ijerph19042282
40. Kannan S, Sheeza A. Omicron (B. 1.1. 529)-variant of concern-molecular profile and epidemiology: a mini review. European Review for Medical and Pharmacological Sciences. 2021 Dec 1;25(24):8019-22. https://doi.org/10.26355/eurrev_202112_27653
41. Chen J, Wei GW. Omicron BA. 2 (b. 1.1. 529.2): High potential for becoming the next dominant variant. The Journal of Physical Chemistry Letters. 2022 May 5;13:3840-9. https://doi.org/10.1021/acs.jpclett.2c00469
42. Gavriatopoulou M, Ntanasis-Stathopoulos I, Korompoki E, Fotiou D, Migkou M, Tzanninis IG, Psaltopoulou T, Kastritis E, Terpos E, Dimopoulos MA. Emerging treatment strategies for COVID-19 infection. Clinical and Experimental Medicine. 2021 May;21(2):167-79. https://doi.org/10.1007/s10238-020-00671-y
43. RECOVERY Collaborative Group. Dexamethasone in hospitalized patients with Covid-19. New England Journal of Medicine. 2021 Feb 25;384(8):693-704. https://doi.org/10.1056/NEJMoa2021436
44. Ali MA, Spinler SA. COVID-19 and thrombosis: From bench to bedside. Trends in Cardiovascular Medicine. 2021 Apr 1;31(3):143-60. https://doi.org/10.1016/j.tcm.2020.12.004
45. Carfora V, Spiniello G, Ricciolino R, Di Mauro M, Migliaccio MG, Mottola FF, Verde N, Coppola N. Anticoagulant treatment in COVID-19: A narrative review. Journal of Thrombosis and Thrombolysis. 2021 Apr;51(3):642-8. https://doi.org/10.1007/s11239-020-02242-0
46. Marik PE, Kory P, Varon J, Iglesias J, Meduri GU. MATH+ protocol for the treatment of SARS-CoV-2 infection: the scientific rationale. Expert Review of Anti-infective Therapy. 2021 Feb 1;19(2):129-35. https://doi.org/10.1080/14787210.2020.1808462
47. Hwang YC, Lu RM, Su SC, Chiang PY, Ko SH, Ke FY, Liang KH, Hsieh TY, Wu HC. Monoclonal antibodies for COVID-19 therapy and SARS-CoV-2 detection. Journal of Biomedical Science. 2022 Dec;29(1):1-50. https://doi.org/10.1186/s12929-021-00784-w
48. Levy JH, Iba T, Olson LB, Corey KM, Ghadimi K, Connors JM. COVID?19: Thrombosis, thromboinflammation, and anticoagulation considerations. International Journal of Laboratory Hematology. 2021 Jul;43:29-35. https://doi.org/10.1111/ijlh.13500
49. Alwan NA, Johnson L. Defining long COVID: Going back to the start. Med. 2021 May 14;2(5):501-4. https://doi.org/10.1016/j.medj.2021.03.003
50. Nuzzo D, Vasto S, Scalisi L, Cottone S, Cambula G, Rizzo M, Giacomazza D, Picone P. Post-acute COVID-19 neurological syndrome: A new medical challenge. Journal of Clinical Medicine. 2021 Jan;10(9):1947. https://doi.org/10.3390/jcm10091947
51. Tortolero GA, de Oliveira Otto M, Ramphul R, Yamal JM, Rector A, Brown M, Peskin MF, Mofleh D, Boerwinkle E. Examining social vulnerability and the association with COVID-19 incidence in Harris County, Texas. Frontiers in Public Health. 2021;9: 798085. https://doi.org/10.3389/fpubh.2021.798085
52. Valerio-Shewmaker MA, Stacia DM, Swartz MD, Yaseen A, Gonzalez MO, Kohl III HW, Kelder SH, Messiah SE, Aguillard KA, Breaux C, Wu L. Strategies to estimate prevalence of SARS-CoV-2 antibodies in a Texas vulnerable population: Results from Phase I of the Texas Coronavirus Antibody REsponse Survey (TX CARES). Frontiers in Public Health. 2021 Dec 14:1947. https://doi.org/10.3389/fpubh.2021.753487
53. Long SW, Olsen RJ, Christensen PA, Bernard DW, Davis JJ, Shukla M, Nguyen M, Saavedra MO, Yerramilli P, Pruitt L, Subedi S. Molecular architecture of early dissemination and massive second wave of the SARS-CoV-2 virus in a major metropolitan area. mBio 11: e02707-20. https://doi.org/10.1128/mBio.02707-20
54. Christensen PA, Olsen RJ, Long SW, Snehal R, Davis JJ, Saavedra MO, Reppond K, Shyer MN, Cambric J, Gadd R, Thakur RM. Signals of significantly increased vaccine breakthrough, decreased hospitalization rates, and less severe disease in patients with Coronavirus disease 2019 caused by the Omicron variant of severe acute respiratory syndrome Coronavirus 2 in Houston, Texas. The American Journal of Pathology. 2022 Apr 1;192(4):642-52. https://doi.org/10.1016/j.ajpath.2022.01.007
55. Christensen PA, Olsen RJ, Long SW, Subedi S, Davis JJ, Hodjat P, Walley DR, Kinskey JC, Saavedra MO, Pruitt L, Reppond K. Delta variants of SARS-CoV-2 cause significantly increased vaccine breakthrough COVID-19 cases in Houston, Texas. The American Journal of Pathology. 2022 Feb 1;192(2):320-31. https://doi.org/10.1016/j.ajpath.2021.10.019
56. Long SW, Olsen RJ, Christensen PA, Subedi S, Olson R, Davis JJ, Saavedra MO, Yerramilli P, Pruitt L, Reppond K, Shyer MN. Sequence analysis of 20,453 severe acute respiratory syndrome coronavirus 2 genomes from the Houston metropolitan area identifies the emergence and widespread distribution of multiple isolates of all major variants of concern. The American Journal of Pathology. 2021 Jun 1;191(6):983-92. https://doi.org/10.1016/j.ajpath.2021.03.004
57. Maccio U, Zinkernagel AS, Schuepbach R, Probst-Mueller E, Frontzek K, Brugger SD, Hofmaenner DA, Moch H, Varga Z. Long-term persisting SARS-CoV-2 RNA and pathological findings: lessons learnt from a series of 35 COVID-19 autopsies. Frontiers in Medicine. 2022;9. https://doi.org/10.3389/fmed.2022.778489
58. Lucas S. Pandemics and pathology: a reflection on influenza, HIV/AIDS and SARS (COVID-19) pandemic infections. Diagnostic Histopathology. 2021 Mar 1;27(3):128-33. https://doi.org/10.1016/j.mpdhp.2020.12.001
59. Hariri LP, North CM, Shih AR, Israel RA, Maley JH, Villalba JA, Vinarsky V, Rubin J, Okin DA, Sclafani A, Alladina JW. Lung histopathology in coronavirus disease 2019 as compared with severe acute respiratory sydrome and H1N1 influenza: A systematic review. Chest. 2021 Jan 1;159(1):73-84. https://doi.org/10.1016/j.chest.2020.09.259
60. Wright FL, Vogler TO, Moore EE, Moore HB, Wohlauer MV, Urban S, Nydam TL, Moore PK, McIntyre Jr RC. Fibrinolysis shutdown correlation with thromboembolic events in severe COVID-19 infection. Journal of the American College of Surgeons. 2020 Aug 1;231(2):193-203. https://doi.org/10.1016/j.jamcollsurg.2020.05.007
61. Attisani L, Pucci A, Luoni G, et al. (2021) COVID-19 and acute limb ischemia: A systematic review. J Cardiovasc Surg (Torino).;62:542-547. https://doi.org/10.23736/S0021-9509.21.12017-8
62. McGonagle D, Bridgewood C, Ramanan AV, Meaney JF, Watad A. COVID-19 vasculitis and novel vasculitis mimics. The Lancet Rheumatology. 2021 Mar 1;3(3):e224-33. https://doi.org/10.1016/S2665-9913(20)30420-3
63. Ammirati E, Lupi L, Palazzini M, Hendren NS, Grodin JL, Cannistraci CV, Schmidt M, Hekimian G, Peretto G, Bochaton T, Hayek A. Prevalence, characteristics, and outcomes of COVID-19–associated acute myocarditis. Circulation. 2022 Apr 12;145(15):1123-39. https://doi.org/10.1161/CIRCULATIONAHA.121.056817
64. Thomson A, Totaro R, Cooper W, Dennis M. Fulminant Delta COVID-19 myocarditis: a case report of fatal primary cardiac dysfunction. European Heart Journal-Case Reports. 2022 Apr;6(4):ytac142. https://doi.org/10.1093/ehjcr/ytac142
65. Verma AK, Lavine KJ, Lin CY. Myocarditis after Covid-19 mRNA vaccination. New England Journal of Medicine. 2021 Sep 30;385(14):1332-4. https://doi.org/10.1056/NEJMc2109975
66. Diaz GA, Parsons GT, Gering SK, Meier AR, Hutchinson IV, Robicsek A. Myocarditis and pericarditis after vaccination for COVID-19. Jama. 2021 Sep 28;326(12):1210-2. https://doi.org/10.1001/jama.2021.13443
67. Patone M, Mei XW, Handunnetthi L, Dixon S, Zaccardi F, Shankar-Hari M, Watkinson P, Khunti K, Harnden A, Coupland CA, Channon KM. Risks of myocarditis, pericarditis, and cardiac arrhythmias associated with COVID-19 vaccination or SARS-CoV-2 infection. Nature Medicine. 2022 Feb;28(2):410-22. https://doi.org/10.1038/s41591-021-01630-0
68. Truong DT, Dionne A, Muniz JC, McHugh KE, Portman MA, Lambert LM, Thacker D, Elias MD, Li JS, Toro-Salazar OH, Anderson BR. Clinically suspected myocarditis temporally related to COVID-19 vaccination in adolescents and young adults. Circulation. 2021 Dec 6 145:345-356. https://doi.org/10.1161/CIRCULATIONAHA.121.056583
69. Heymans S, Cooper LT. Myocarditis after COVID-19 mRNA vaccination: clinical observations and potential mechanisms. Nature Reviews Cardiology. 2021 Dec 9:1-3. https://doi.org/10.1038/s41569-021-00662-w
70. Raman B, Bluemke DA, Lüscher TF, Neubauer S. Long COVID: post-acute sequelae of COVID-19 with a cardiovascular focus. European Heart Journal. 2022 Mar 14;43(11):1157-72. https://doi.org/10.1093/eurheartj/ehac031
71. Hugon J, Msika EF, Queneau M, Farid K, Paquet C. Long COVID: cognitive complaints (brain fog) and dysfunction of the cingulate cortex. Journal of Neurology. 2022 Jan;269(1):44-6. https://doi.org/10.1007/s00415-021-10655-x
72. Mehandru S, Merad M. Pathological sequelae of long-haul COVID. Nature Immunology. 2022 Feb;23(2):194-202. https://doi.org/10.1038/s41590-021-01104-y
73. Theoharides TC. Could SARS-CoV-2 Spike Protein Be Responsible for Long-COVID Syndrome?. Molecular Neurobiology. 2022 Jan 59:1850-1861. https://doi.org/10.1007/s12035-021-02696-0
74. Merad M, Blish CA, Sallusto F, Iwasaki A. The immunology and immunopathology of COVID-19. Science. 2022 Mar 11;375(6585):1122-7. https://doi.org/10.1126/science.abm8108
75. Korompoki E, Gavriatopoulou M, Hicklen RS, Ntanasis-Stathopoulos I, Kastritis E, Fotiou D, Stamatelopoulos K, Terpos E, Kotanidou A, Hagberg CA, Dimopoulos MA. Epidemiology and organ specific sequelae of post-acute COVID19: A narrative review. Journal of Infection. 2021 Jul 1;83(1):1-6. https://doi.org/10.1016/j.jinf.2021.05.004
76. Desai AD, Lavelle M, Boursiquot BC, Wan EY. Long-term complications of COVID-19. American Journal of Physiology-Cell Physiology. 2022 Jan 1;322(1):C1-1. https://doi.org/10.1152/ajpcell.00375.2021
77. Rutkai I, Mayer MG, Hellmers LM, Ning B, Huang Z, Monjure CJ, Coyne C, Silvestri R, Golden N, Hensley K, Chandler K. Neuropathology and virus in brain of SARS-CoV-2 infected non-human primates. Nature Communications. 2022 Apr 1;13(1): 1745. https://doi.org/10.1038/s41467-022-29440-z
78. Subbaraman N. US health agency will invest $1 billion to investigate'long COVID'. Nature. 2021 Mar 18;591(7850):356-7. https://doi.org/10.1038/d41586-021-00586-y